| CONDENSED MATTER: STRUCTURE, MECHANICAL AND THERMAL PROPERTIES |
 |
|

|
|
|
Unexpected Selective Absorption of Lithium in Thermally Reduced Graphene Oxide Membranes |
| Jie Jiang1,2†, Liuhua Mu1,2†, Yu Qiang3†, Yizhou Yang3, Zhikun Wang4, Ruobing Yi1,2, Yinwei Qiu4, Liang Chen4*, Long Yan1*, and Haiping Fang1,3* |
1Shanghai Institute of Applied Physics, Chinese Academy of Sciences, Shanghai 201800, China
2University of Chinese Academy of Sciences, Beijing 100049, China
3Department of Physics, East China University of Science and Technology, Shanghai 200237, China
4Department of Optical Engineering, Zhejiang Provincial Key Laboratory of Chemical Utilization of Forestry Biomass, Zhejiang A&F University, Hangzhou 311300, China
|
|
| Cite this article: |
|
Jie Jiang, Liuhua Mu, Yu Qiang et al 2021 Chin. Phys. Lett. 38 116802 |
|
|
|
|
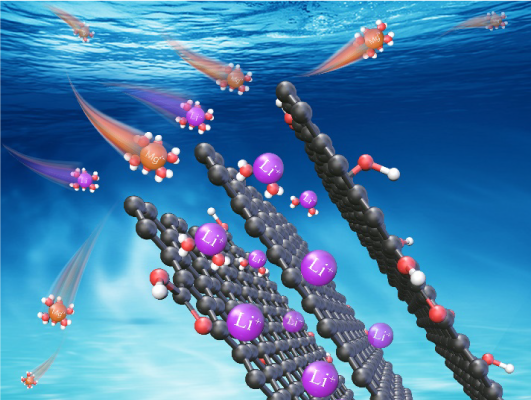
Abstract Lithium plays an increasingly important role in scientific and industrial processes, and it is extremely important to extract lithium from a high Mg$^{2+}$/Li$^{+}$ mass ratio brine or to recover lithium from the leachate of spent lithium-ion batteries. Conventional wisdom shows that Li$^{+}$ with low valence states has a much weaker adsorption (and absorption energy) with graphene than multivalent ions such as Mg$^{2+}$. Here, we show the selective adsorption of Li$^{+}$ in thermally reduced graphene oxide (rGO) membranes over other metal ions such as Mg$^{2+}$, Co$^{2+}$, Mn$^{2+}$, Ni$^{2+}$, or Fe$^{2+}$. Interestingly, the adsorption strength of Li$^{+}$ reaches up to 5 times the adsorption strength of Mg$^{2+}$, and the mass ratio of a mixed Mg$^{2+}$/Li$^{+}$ solution at a very high value of $ 500\!:\!1$ can be effectively reduced to $ 0.7\!:\!1$ within only six experimental treatment cycles, demonstrating the excellent applicability of the rGO membranes in the Mg$^{2+}$/Li$^{+}$ separation. A theoretical analysis indicates that this unexpected selectivity is attributed to the competition between cation–$\pi$ interaction and steric exclusion when hydrated cations enter the confined space of the rGO membranes.
|
|
Received: 29 September 2021
Express Letter
Published: 27 October 2021
|
|
|
| Fund: Supported by the Fundamental Research Funds for the Central Universities, the National Natural Science Foundation of China (Grant Nos. 11974366, 11675246, 12074341, U1832170, and U1832150), the Key Research Program of Chinese Academy of Sciences (Grant No. QYZDJ-SSW-SLH053), the Computer Network Information Center of the Chinese Academy of Sciences, and the Shanghai Supercomputer Center of China. |
|
|
|
|
Viewed |
|
|
|
Full text
|
|
|
|
|
Abstract
|
|
|
|
|