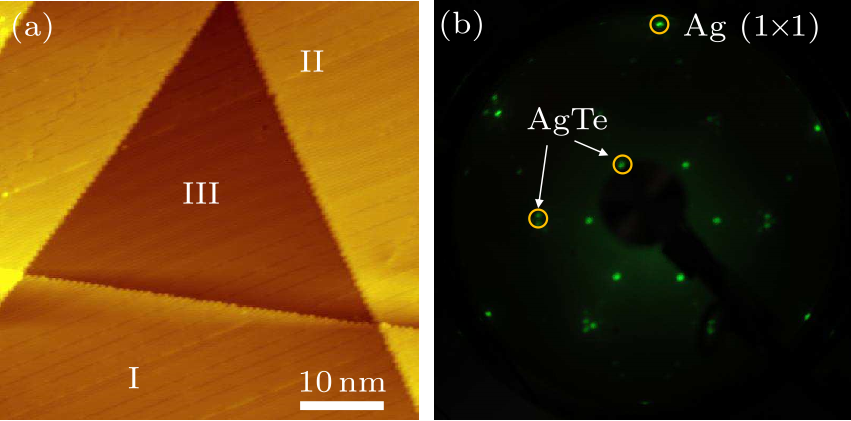
Fig. 1. (a) STM image of large-scale AgTe monolayer on Ag(111) substrate. Scanning parameters: $V_{\rm s}=-0.6$ V, $I_{\rm t}=0.2$ nA. (b) LEED pattern of the sample in (a), with a beam energy of 55.4 eV.
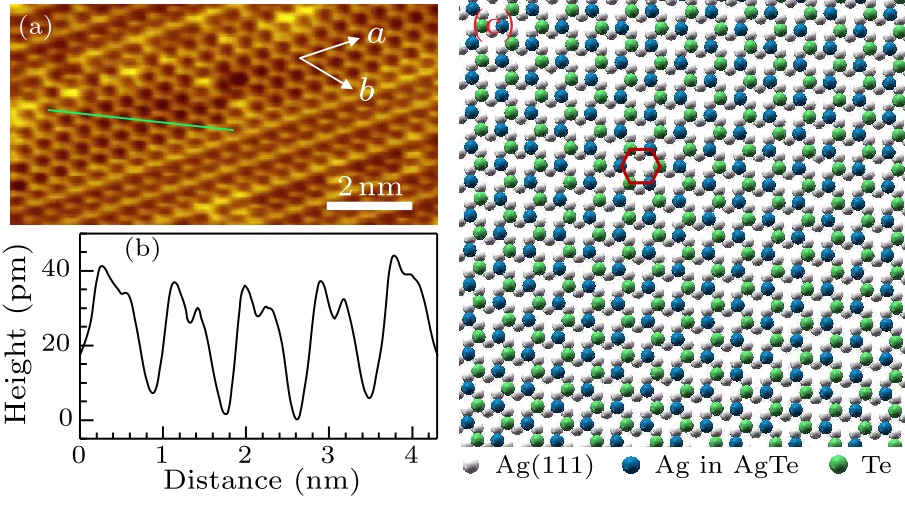
Fig. 2. (a) STM image of monolayer AgTe showing the flat honeycomb regions. Scanning parameters: $V_{\rm s}=-0.6$ V, $I_{\rm t}=0.1$ nA. (b) Line profile of the honeycomb structure showing the asymmetric sublattices. (c) Schematic of the flat honeycomb structure of AgTe on Ag(111) substrate.
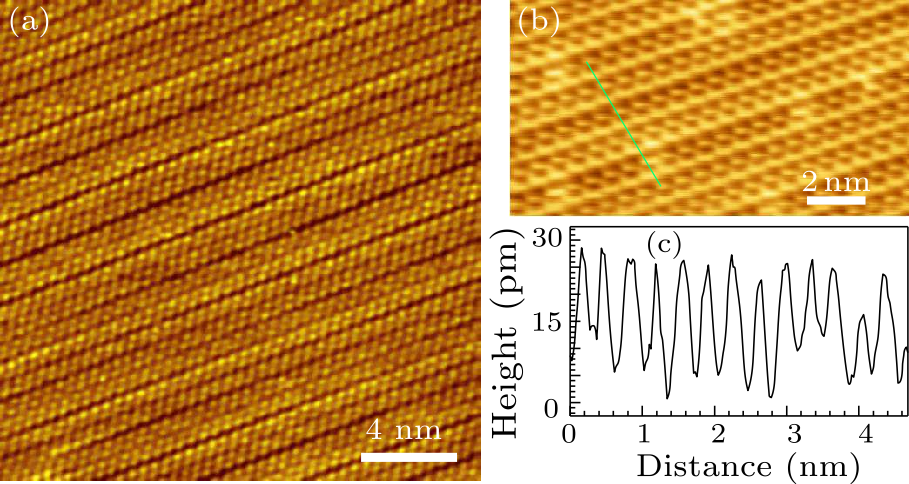
Fig. 3. (a) STM image of the buckled honeycomb regions. Scanning parameters: $V_{\rm s}=-1.5$ V, $I_{\rm t}=0.4$ nA. (b) Zoom-in image of (a) showing the buckled honeycomb structure. Scanning parameters: $V_{\rm s}=-0.6$ V, $I_{\rm t}=0.1$ nA. (c) Line profile of the buckled honeycomb structure.

Fig. 4. (a) STM image showing the stripe structure. Scanning parameters: $V_{\rm s}=-1.8$ V, $I_{\rm t}=0.8$ nA. (b) Schematic of the stripe formation of the AgTe on Ag(111) substrate.
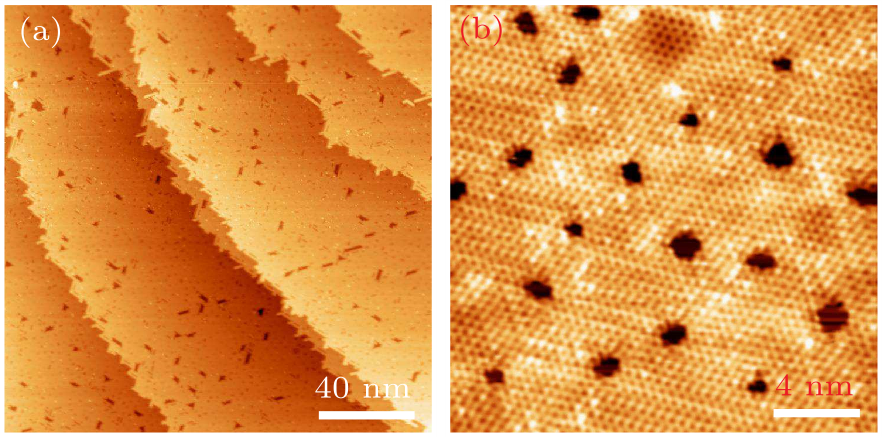
Fig. 5. (a) Large-scale and (b) atomic resolution STM images of the AgTe on Ag(111) with higher Te coverage, showing the patterned hexagonal structure of AgTe. Scanning parameters: $V_{\rm s}=-1.8$ V, $I_{\rm t}=0.8$ nA.
| [1] | Novoselov K S, Geim A K, Morozov S V, Jiang D, Zhang Y, Dubonos S V, Grigorieva I V and Firsov A A 2004 Science 306 666 | Electric Field Effect in Atomically Thin Carbon Films
| [2] | Novoselov K S, Mishchenko A, Carvalho A and Neto A H C 2016 Science 353 aac9439 | 2D materials and van der Waals heterostructures
| [3] | Pan Y, Zhang L Z, Huang L, Li L F, Meng L, Gao M, Huan Q, Lin X, Wang Y L, Du S X, Freund H J and Gao H J 2014 Small 10 2215 | Construction of 2D Atomic Crystals on Transition Metal Surfaces: Graphene, Silicene, and Hafnene
| [4] | Zhang G Y, Du S X, Wu K H and Gao H J 2018 Science 360 673 | Sponsored Collection | Humble Beginning, Bright Future: Institute of Physics (CAS) at 90
| [5] | Li G, Zhang Y Y, Guo H, Huang L, Lu H L, Lin X, Wang Y L, Du S X and Gao H J 2018 Chem. Soc. Rev. 47 6073 | Epitaxial growth and physical properties of 2D materials beyond graphene: from monatomic materials to binary compounds
| [6] | Zhang S L, Guo S Y, Chen Z F, Wang Y L, Gao H J, Gomez-Herrero J, Ares P, Zamora F, Zhu Z and Zeng H B 2018 Chem. Soc. Rev. 47 982 | Recent progress in 2D group-VA semiconductors: from theory to experiment
| [7] | Pan Y, Shi D X and Gao H J 2007 Chin. Phys. 16 3151 | Formation of graphene on Ru(0001) surface
| [8] | Pan Y, Zhang H G, Shi D X, Sun J T, Du S X, Liu F and Gao H J 2009 Adv. Mater. 21 2777 | Highly Ordered, Millimeter-Scale, Continuous, Single-Crystalline Graphene Monolayer Formed on Ru (0001)
| [9] | Huang L, Xu W Y, Que Y D, Mao J H, Meng L, Pan L D, Li G, Wang Y L, Du S X, Liu Y Q and Gao H J 2013 Chin. Phys. B 22 096803 | Intercalation of metals and silicon at the interface of epitaxial graphene and its substrates
| [10] | Meng L, Wang Y L, Zhang L Z, Du S X, Wu R T, Li L F, Zhang Y, Li G, Zhou H T, Hofer W A and Gao H J 2013 Nano Lett. 13 685 | Buckled Silicene Formation on Ir(111)
| [11] | Huang L, Zhang Y F, Zhang Y Y, Xu W Y, Que Y D, Li E, Pan J B, Wang Y L, Liu Y Q, Du S X, Pantelides S T and Gao H J 2017 Nano Lett. 17 1161 | Sequence of Silicon Monolayer Structures Grown on a Ru Surface: from a Herringbone Structure to Silicene
| [12] | Li G, Zhang L Z, Xu W Y, Pan J B, Song S R, Zhang Y, Zhou H T, Wang Y L, Bao L H, Zhang Y Y, Du S X, Ouyang M, Pantelides S T and Gao H J 2018 Adv. Mater. 30 1804650 | Stable Silicene in Graphene/Silicene Van der Waals Heterostructures
| [13] | Li L F, Lu S Z, Pan J B, Qin Z H, Wang Y Q, Wang Y L, Cao G Y, Du S X and Gao H J 2014 Adv. Mater. 26 4820 | Buckled Germanene Formation on Pt(111)
| [14] | Wu X, Shao Y, Liu H, Feng Z, Wang Y L, Sun J T, Liu C, Wang J O, Liu Z L, Zhu S Y, Wang Y Q, Du S X, Shi Y G, Ibrahim K and Gao H J 2017 Adv. Mater. 29 1605407 | Epitaxial Growth and Air-Stability of Monolayer Antimonene on PdTe 2
| [15] | Shao Y, Liu Z L, Cheng C, Wu X, Liu H, Liu C, Wang J O, Zhu S Y, Wang Y Q, Shi D X, Ibrahim K, Sun J T, Wang Y L and Gao H J 2018 Nano Lett. 18 2133 | Epitaxial Growth of Flat Antimonene Monolayer: A New Honeycomb Analogue of Graphene
| [16] | Zhu F F, Chen W J, Xu Y, Gao C L, Guan D D, Liu C H, Qian D, Zhang S C and Jia J F 2015 Nat. Mater. 14 1020 | Epitaxial growth of two-dimensional stanene
| [17] | Zhang Z H, Penev E S and Yakobson B I 2017 Chem. Soc. Rev. 46 6746 | Two-dimensional boron: structures, properties and applications
| [18] | Wang Q H, Kalantar-Zadeh K, Kis A, Coleman J N and Strano M S 2012 Nat. Nanotechnol. 7 699 | Electronics and optoelectronics of two-dimensional transition metal dichalcogenides
| [19] | Fatemi V, Wu S F, Cao Y, Bretheau L, Gibson Q D, Watanabe K, Taniguchi T, Cava R J and Jarillo-Herrero P 2018 Science 362 926 | Electrically tunable low-density superconductivity in a monolayer topological insulator
| [20] | Lin X, Lu J C, Shao Y, Zhang Y Y, Wu X, Pan J B, Gao L, Zhu S Y, Qian K, Zhang Y F, Bao D L, Li L F, Wang Y Q, Liu Z L, Sun J T, Lei T, Liu C, Wang J O, Ibrahim K, Leonard D N, Zhou W, Guo H M, Wang Y L, Du S X, Pantelides S T and Gao H J 2017 Nat. Mater. 16 717 | Intrinsically patterned two-dimensional materials for selective adsorption of molecules and nanoclusters
| [21] | Gao L, Sun J T, Lu J C, Li H, Qian K, Zhang S, Zhang Y Y, Qian T, Ding H, Lin X, Du S and Gao H J 2018 Adv. Mater. 30 1707055 | Epitaxial Growth of Honeycomb Monolayer CuSe with Dirac Nodal Line Fermions
| [22] | Anasori B, Lukatskaya M R and Gogotsi Y 2017 Nat. Rev. Mater. 2 16098 | 2D metal carbides and nitrides (MXenes) for energy storage
| [23] | Wang Y, Xiao J, Zhu H Y, Li Y, Alsaid Y, Fong K Y, Zhou Y, Wang S Q, Shi W, Wang Y, Zettl A, Reed E J and Zhang X 2017 Nature 550 487 | Structural phase transition in monolayer MoTe2 driven by electrostatic doping
| [24] | Zhang Y C and Stucky G D 2014 Chem. Mater. 26 837 | Heterostructured Approaches to Efficient Thermoelectric Materials
| [25] | van Veggel F C J M 2014 Chem. Mater. 26 111 | Near-Infrared Quantum Dots and Their Delicate Synthesis, Challenging Characterization, and Exciting Potential Applications
| [26] | Zhang W, Yu R, Feng W X, Yao Y G, Weng H M, Dai X and Fang Z 2011 Phys. Rev. Lett. 106 156808 | Topological Aspect and Quantum Magnetoresistance of
| [27] | Ma Y D, Kou L Z, Dai Y and Heine T 2016 Phys. Rev. B 93 235451 | Two-dimensional topological insulators in group-11 chalcogenide compounds:
| [28] | Zhang Y H, Li Y, Ma Y M, Li Y W, Li G H, Shao X C, Wang H, Cui T, Wang X and Zhu P W 2015 Sci. Rep. 5 14681 | Electronic Topological Transition in Ag2Te at High-pressure
| [29] | Sáfrán G, Geszti O, Radnóczi G 2003 Vacuum 71 299 | Formation of oriented silver telluride on single and polycrystalline Ag films